Written by: Tianna Dupont, WSU Tree Fruit Extension. January 2017; edited October 2018.
Introduction
WSU has a long history of soils research dating back to the university’s establishment in 1892. Today’s researchers strive to improve soil nutrient use efficiency while increasing tree fruit quality using sustainable soil management and precision technologies. More information about current soil research and practices can be found at the Organic and Integrated Tree Fruit Production webpage on soils. Please also see our Orchard Establishment and Organic Production pages for more soils related information.
What is soil?
Soil is a complex, living and breathing system composed of living, decomposing and dead matter, all interacting with the physical and chemical properties of the soil. When the biologically interrelated organisms in the soil food web are in balance and are able to support plant productivity, they create a “healthy” soil. Soil health, also referred to as soil quality, is defined as the continued capacity of soil to function as a vital living ecosystem that sustains plants, animals, and humans. (reference: USDA Natural Resources Conservation Service [NRCS])
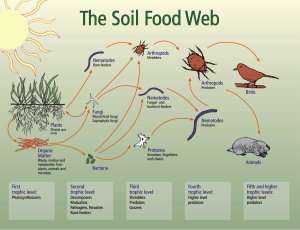
A healthy soil food web (see figure right) is vital to fruit tree nutrition. Indicators of soil quality are structure, water holding capacity, infiltration, pH, soil organic matter, and microbial activity. Every location on earth has a unique soil and every type of crop requires a specific balance of nutrients and soil chemistry for optimal plant health. Fruit trees in North Central Washington require soil with adequate structure and permeability to circulate water and air to the tree roots, as well as providing anchorage for tree stability. Orchard blocks will often have variations in soil types within the blocks. Preferred orchard soil is a stable, sandy loam soil that is granular and porous, enabling good water and air movement. Soil pH around 6.0 to 7.5, slightly acidic, is optimal for fruit trees to absorb nutrients.
There are certain soil conditions that are detrimental to the health of fruit trees. Replant disease, nutrient excess or deficiencies, poor soil texture, poor water holding capacity or poor water drainage (e.g., waterlogging) are all important soil issues. Soil at new or replant sites should be tested for pH, soil nutrient availability, and the presence of harmful nematodes. Replant disease is a tree condition involving microorganisms in the soil that attack and damage tree roots, leading to poor tree growth and productivity. Soil pathogens contributing to replant disease are fungi, bacteria, viruses, and nematodes. These microorganisms can build up in the soil rhizosphere over the years. When old trees are removed, microorganisms remain in soil and roots left behind. Conventional orchardists typically fumigate the replant site after the trees and root debris have been removed. It is important to remove as much of the old root debris as possible before fumigating to be effective. There are alternatives to fumigation for preventing replant disease. More information can be found on controlling replant disease at our Orchard Establishment page.
Properties of Soil
Physical Properties
Soil permeability is how fast water moves through the soil. Macropores control a soil’s permeability and aeration. The Water Holding Capacity (WHC) of soil is the soil’s ability to hold water for plant use. Micropores are responsible for a soil’s WHC. Different types of soil (sand, silt, and clay) have different pore sizes, which allows water to drain through soil at different rates. The proportions of sand, silt and clay in soil determine the water holding capacity. Medium textured soils (silt/loams) are the most suitable for plant growth. Soil texture, soil structure, compaction, and organic matter all affect soil porosity.
Soil texture overview:
- Relative soil particle sizes: sand (coarse) > silt > (medium) > clay (fine)
- Sands are the largest particles and feel gritty
- Silts are medium-sized particles and feel soft, silky or floury
- Clays are the smallest sized particles, feel sticky and are hard to squeeze
- Fine-textured soils (high clay content) hold more water than coarse-textured soils but may not be ideal due to low oxygen holding capacity (aeration)
- Course textured soils have high permeability and low WHC making it suitable for only well-adapted plant species
- Medium-textured soils (loam family) are suitable for growth of most plants
Soil structure overview:
Structure refers to the arrangement of aggregated soil particles and determines the soil particle surface area (Table 1).
Table 1. Properties of soil particle size
| Sand | Silt | Clay | |
| Porosity | mostly large pores | small pores predominate | small pores predominate |
| Permeability | rapid | low to moderate | slow |
| Water holding capacity | limited | medium | very large |
| Soil particle surface | small | medium | very large |
Soil Compaction destroys the quality of the soil because it restricts rooting depth and decreases pore size. When soil is compacted, there are more water-filled pores that are less able to absorb water, increasing runoff and erosion, and lowering soil temperatures. To reduce compaction:
- Add organic matter
- Make fewer trips across area with machinery
- Practice reduced-till or no-till systems
- Avoid waterlogging soils with irrigation and harvesting when soils are wet
Chemical Properties
The major chemical properties affecting soils are: soil pH, salinity, cation exchange capacity (CEC), organic matter, and the C:N ratio (Carbon to Nitrogen)
pH
pH measures the acidity or alkalinity of a soil. Neutral = 7.0, Acidic < 7.0, and Alkaline is > 7.0. Soil pH affects the availability of plant nutrients (in general, the optimal pH levels for orchard soil is between 6.0 to 7.5). Soil pH is one of the most indicative measurements of the general chemical status of soil. Because soil pH is typically measured as soil-solution pH, it is also an indicator of the proportions of basic and acidic exchangeable ions present in the soil. This is because these ions in the soil solution are in equilibrium with the exchangeable ions. (see also Cation-exchange capacity). Additionally, pH affects the abundance of microorganisms present in the soil. Bacteria are generally more prevalent in alkaline soils and fungi dominate in acidic soils. This is important because microbes are responsible for the cycling of nutrients. The most diverse and numerous populations of microorganisms are found in near-neutral soils. Furthermore, soil pH influences pathogenic microbes, and growers can adjust pH to manage some plant diseases.
Soil salinity
There can be high salinity problems in irrigated soils due to the high evaporation rates and low annual rainfall that occur leaving salts to accumulate. Severe accumulation of salts may restrict growth.
Cation-exchange capacity (CEC)
CECis defined as the degree to which a soil can adsorb and exchange cations.
- Cation: a positively charged ion (e.g., NH4+, K+, Ca2+, Fe2+, etc.)
- Anion: a negatively charged ion (e.g., NO3-, PO42-, SO42-, etc.)
Soil particles and organic matter have negative charges on their surfaces. Mineral cations can adsorb to the negative surface charges of the inorganic and organic soil particles. Once adsorbed, these minerals are not easily lost when the soil is leached by water and they also provide a nutrient reserve available to plant roots. These minerals can then be replaced or exchanged by other cations (i.e., cation exchange). CEC is highly dependent upon soil texture and organic matter content. In general, the more clay and organic matter in the soil, the higher the CEC. Clay content is important because these small particles have a high surface area to volume ratio.
Plant roots also possess cation exchange capacity. Hydrogen ions from the root hairs and microorganisms may replace nutrient cations from the exchange complex on soil colloids. The nutrient cations are then released into the soil solution where they can be taken up by the adsorptive surfaces of roots and soil organisms. They may, however, be lost from the system by water drainage.
Additionally, high levels of one nutrient may influence uptake of another nutrient (e.g., antagonistic relationships). For example, potassium (K) uptake by plants is limited by high levels of calcium (Ca) in some soils. High levels of potassium (K) can, in turn, limit magnesium (Mg) uptake by the plant, even if Mg levels in soil are high.
Soil Moisture factor
The goal of irrigation is to recharge the available water in the top foot of soil where the feeder roots live and most of the tree water uptake occurs. The amount to irrigate depends on the soil water-holding capacity, weather, the site, and the plants grown and their stage of development. See the Irrigation Management section of this website for information on soil moisture.
- On average, plants consume from 0.1 to 0.3 in of rainfall or irrigation per day
- Sandy soils: Hold between 0.5-1 in. of water per ft. soil
- Loams: Hold between 0.8-2 in. per ft. soil
- Clay: Hold between 1.3-2.4 in. per ft. of soil
Biological Properties
Organic matter has a combination of physical and chemical properties, similar to the soil particles. However, its biological components are of the most importance. Microorganisms are the driving force for nutrient release to plants. Nutrients in the soil organic matter are released to plants more during warm, moist conditions, and slower in cool, dry climates. The flora comprising the organic portion of soil include bacteria, fungi, algae, mosses, vascular plants. Soil fauna can be broken into three major types: Macrofauna, which includes vertebrates (mice, etc. that tunnel and/or nest in the soil), worms, insects and spiders; Mesofauna, which include nematodes, insects, mites and mollusks (snails and slugs); and Microfauna, which include various protozoa (single celled organisms). The combination of these soil organisms are responsible for the cycling of carbon (C), nitrogen (N) and other nutrients. They enhance soil structure, decompose organic matter, increase aeration and penetrability, and are involved in both disease transmission and control.
Soil Sampling
Proper soil sampling is critical in assessing the soil fertility of your site. Soils are naturally variable; therefore, it is necessary to obtain a random and representative sample. Restrict the area sampled to a uniform soil type or condition within the orchard. Collect samples from within the existing tree row and not the grass alleyway. Samples should also be taken from within the drip-line of the tree canopy. Sample topsoil and subsoil separately. Scrape away the surface soil and then collect samples from the 0-6” depth. Sample from the 6”-18” depth, and keep these samples separate from the 0-6” samples. For more information about soil testing visit the WSU Soils and Soil Testing webpage.
Resources:
Webpages
- Drought Website, WSU Extension
- Cover Crops, WSU Organic and Integrated Tree Fruit Production
- Compost, WSU Center for Sustaining Agriculture and Natural Resources
- Soils, Compost and Mulch, WSU Gardening in Washington State.
- Soil sampling, Sallato, WSU.
- Nutrient management in Tree Fruit, Sallato et al., WSU
Videos
- Root health, Fertility, Water and Crop load. B. Sallato (WSU), 2023
- The Hows and Whys of Soil Testing, J. Davenport, (WSU-IAREC), 2014.
- The Potential for Precision Soils Management in Orchards, D. Brown, (WSU-Pullman), 2014.
- Root Physiology and Function in Orchards, L. Kalcsits, (WSU-TFREC), 2014.
- Optimal Tree Nutrition and Fruit Production Begins Underground, G. Fazio, (USDA-ARS), 2014.
- Characteristics of Tree Root Systems, D. Eissenstat, Pennsylvania State University, 2014.
- Roots and Soil Biology: Managing the “Microherd” for Max Tree Performance, M. Mazzola, (USDA-ARS), 2014.
- The Sponge Analogy for Soil Moisture, C. Cogger, (WSU-Puyallup), 2014.
- Soil Sampling, C. Cogger, (WSU-Puyallup).
Presentations
- Soil Health in Orchards: An Introduction, D. Granatstein (WSU-CSANR).
- Linking fruit and soil quality (Health), P. Andrews (WSU-Horticulture), IFTA conference, 2011.
- Understanding Soil Tests, C. Cogger (WSU-Puyallup), 2014.
Trade Articles
- New tests measure soil health, M. Hansen, Good Fruit Grower, April, 2014.
- Pay attention to the soil, G. Warner, Good Fruit Grower, April, 2014.
- New biofumigant registered, M. Hansen, Good Fruit Grower, April, 2014.
