By Achour Amiri, WSU Plant Pathology. Updated April 2020. Downloadable pdf Phacidiopycnis
Phacidiopycnis rots are emerging postharvest diseases of apple and pear in the U.S. Pacific Northwest. These diseases have previously been reported in Europe and India and most recently in Chile. In Washington State and Oregon, Phacidiopycnis rot is common on pears, whereas speck rot is more common on apples in Washington State. Recent surveys have confirmed that these rots continue to be an emerging threat in the PNW and will require future management attention.
Casual Organism
Two Phacidiopycnis species are known to cause Phacidiopycnis rot on apple and pear. Phacidiopycnis rot of pear is caused by is the discomycete Potebniamyces pyri (Berk. & Br.) Dennis, anamorph (asexual) Phacidiopycnis pyri (Fuckel) Weindlmayr. The fungus produces macroconidia and microconidia with the later considered spermatia (male spores) in the fungus life cycle. Conidia germinate either by developing germ tubes, when enough nutrients are available, or by budding to produce secondary conidia when nutrient levels are low.
On apples, Phacidiopycnis rot, more commonly known as speck rot, is caused by Phacidiopycnis washingtonensis (C.L. Xiao & J.D. Rogers) which was first reported in 2005 in the PNW. This species has since been reported on apples in Europe and Chile. Phacidiopycnis washingtonensis grows slightly faster than P. pyri and both species can infect the other fruit but are more common on their respective hosts, apple and pear.
Symptoms
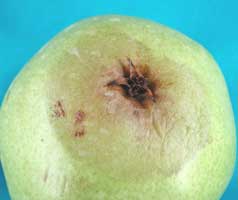
Phacidiopycnis species cause infections via three different pathways: stem-end, calyx-end, or through wounds causing respectively stem-end rot, calyx-end rot and wound rot, respectively (Fig. 1). Decayed area is spongy. In the early stage of symptom development, the decayed area appears water-soaked. Color of the decayed area varies with age. As the disease progresses, the advanced decayed area turns brown and then black, but the margin of decayed area continues to have a water-soaked appearance. Under high relative humidity, the fungus forms white mycelium. Pycnidia (pimple shaped fruiting bodies) of the fungus often form on the decayed area at advanced stages, starting from the infection sites.
In the early stage of symptom development, Phacidiopycnis and speck rots can be misdiagnosed as gray mold or Sphaeropsis rot. The fruit flesh at the margin of Phacidiopycnis rots appears translucent and water-soaked, whereas internal decayed flesh of gray mold usually appears brown (Table 1). Speck rot is slightly firmer than gray mold and forms brown to black specks around lenticels, especially on red apple cultivars.
Figure 1. Symptoms and signs of Phacidiopycnis and Speck rot infections on pome fruit
Photo credit: Achour Amiri (WSU-TFREC)
Internal view of a rare speck rot infection on Fuji in an orchards in the Pacifc Northwest after a heavy rain.
Cross-section of a speck rot lesion showing black coloration of the internal flesh of a Granny Smith apple.
Table 1. Comparison between symptoms of Phacidiopycnis rot & Speck rots and gray mold on pears
| Characteristics | Gray Mold | Speck & Phacidiopycnis rots | Spheropsis rot |
|---|---|---|---|
| Texture | Spongy early, softer as decay advances, decayed tissue not separable from healthy tissue | Spongy slightly firmer than gray mold, decayed tissue not separable from healthy | Firm |
| Color of decayed area | Light to dark brown | Water-soaked, brown then turns black and fruit stays spongy, Brown to black specks | Brown to dark brown, advanced decayed area may turn black |
| Mycelia and spores on fruit | Fluffy white-gray mycelia, gray to brown spore masses under high humidity | White mycelia, rare appearance of black pycnidia at advanced decay stages | At advanced stages and under high humidity white mycelia and pycnidia may form on decayed fruit |
| Color of internal flesh | Light brown to brown | Brown to black | Brown, decay advances along the vascular tissue of the fruit |
| Odor | Not detectable generally, Cider-like on very decayed fruit | Mild, distinct | Wild-strong, bandage-like odor |
Infection and disease cycle of Phacidiopycnis species
Phacidiopycnis pyri and P. washingtonensis are weak canker-causing pathogens on trees. Infection of fruit by these two pathogens occurs exclusively in the orchard, and symptoms develop during storage. Phacidiopycnis pyri is commonly associated with dead bark and cankers on pear trees and survives as mycelium and pycnidia, the primary inoculum for fruit infection, on diseased twigs or dead bark. P. pyri infects the stem and calyx of pear fruit as well as wounds caused by insects, limb rubs or harvest punctures. The stem and calyx of the fruit are more susceptible to infection by the fungus near harvest. Infections occur with warm wet conditions (optimum 68-77 ºF, some growth 26-77ºF, little above 86ºF). Four to 8 hours of wetness duration at optimum temperature are required for infections. Wound infections develop decay symptoms within two months after harvest whereas, stem-end and calyx-end rots symptoms are usually first seen three months or more after harvest.
Under high relative humidity, the fungus produces fluffy mycelium on decayed fruit, which can infect surrounding healthy fruit, causing secondary infections (nests) through fruit-to-fruit spreading.
Phacidiopycnis washingtonensis is also associated with cankers and twig dieback on apple trees and crabapple pollinizers. After maturation, pycnidia are the main primary source of inoculum. Although P. washingtonensis can infect the fruit through wounds, infections through the stem-end and calyx-end are more common in the orchard. On rare occasions, infections via lenticels may occur on over-mature fruit showing lenticel breakdown. Like gray mold and P. pyri, speck rot can infect surrounding healthy fruit, causing secondary infections (nests) through fruit-to-fruit contact. The fungus can grow and germinate between -3 to 25ºC (26-77ºF) with optimum growth occurring between 15-20ºC (59-68ºF). Exact wetness duration requirement is not known, but rain preceding harvest increases infection risk and eventually speck rot incidence in storage.
Cultural Controls
Since both Phacidiopycnis species are associated with cankers and dieback, preharvest sanitation practices are key in managing Phacidiopycnis rots. On pear, good pruning and removal of pruned material is recommended to reduce the inoculum load available in the upcoming season. Inoculum of Phacidiopycnis washingtonensis comes mainly from crabapple pollinizers, therefore, a good pruning along with a fungicide treatment of the pollinizers is key in managing the decay preharvest.
Chemical Controls
Preharvest fungicide applications may prevent infections by Phacidiopycnis species. Among the current preharvest fungicides, Topsin-M has been found to be more effective compared to Pristine. Ziram applied within two weeks before harvest provides some control of stem-end and calyx-end rots. Because these two pathogens infect fruit through the stem- and calyx-ends, it is important to ensure good spray coverage of these potential entries to minimize such infections. Recent research has shown that current postharvest fungicides are the most effective in managing both Phacidiopycnis species with fludioxonil (Scholar) being the most effective (Tables 2 and 3). Difenoconazole (Academy) has been labeled for postharvest application on pome fruit in 2016 and has shown a strong efficacy, especially against P. pyri. Formulations to apply thiabendazole, pyrimethanil and fludioxonil through thermonebulization (fog) exist already. In Washington State, Academy can be applied via drench at harvest if packers can follow specific fungicide waste management practices. A formulation for a dry application of Academy is under development. Phacidiopycnis species have a medium risk for fungicide resistance development, therefore, rotation of fungicides from different FRAC groups in the orchards and warehouse is important to avoid selection for populations that are resistant to current fungicides.
Table 2. List and efficacy of preharvest fungicides registered to control Phacidiopycnis and other decay rots
| Active Ingredient | Trade names | FRAC group | Rate/ac | PHI (days) | Registered for Apple | Registered for Pear | Efficacyb against gray mold |
|---|---|---|---|---|---|---|---|
| Thiophanate-methyl | Topsin-M 70WSB | 1 | 0.75-1 lb | 1 | + | + | Good |
| Boscalid | Pristine | 7 | 18.5 oz | 0 | + | + | Fair |
| Fluxapyroxad | Merivon | 7 | 5.5 fl oz | 0 | + | + | Unknown |
| Pyraclostrobin | Flint Extra, Pristine, Merivon | 11 | see labels | 0 | – | + | Fair |
| Fenhexamid | Elevate 50WDG | 17 | 1-1.5 lb | 0 | – | + | NA |
| Ziram | Ziram 76DF | M03 | 6 lb | 14 | + | + | Fair |
| Captan | Captan 50WP | M04 | 6 lb/ 20-400 gal | 0 | + | – | Fair |
Table 3. List and efficacy of postharvest fungicides registered to control Phacidiopycnis and other decay rots
| Active Ingredient | Trade Names | FRAC group | Rate | PHI (days) | Registered for Apple | Registered for Pear | Efficacy against Phacidiopycnis rots |
|---|---|---|---|---|---|---|---|
| Thiabendazole | TBZ, Mertect, eFog-100 | 1 | see label | NA | + | + | Good |
| Pyrimethanil | Penbotec, eFog 160 | 9 | see label | NA | + | + | Very Good |
| Fludioxonil | Scholar, eFog 80, Actimist | 12 | see label | NA | + | + | Excellent |
| Diffenoconazole + fludioxonil |
Academy | 3 + 12 | see label | NA | + | + | Excellent |
| Captan | Captec 4L | M04 | see label | NA | + | + | Fair |
Use pesticides with care. Apply them only to plants, animals, or sites listed on the labels. When mixing and applying pesticides, follow all label precautions to protect yourself and others around you. It is a violation of the law to disregard label directions. If pesticides are spilled on skin or clothing, remove clothing and wash skin thoroughly. Store pesticides in their original containers and keep them out of the reach of children, pets, and livestock.
YOU ARE REQUIRED BY LAW TO FOLLOW THE LABEL. It is a legal document. Always read the label before using any pesticide. You, the grower, are responsible for safe pesticide use. Trade (brand) names are provided for your reference only. No discrimination is intended, and other pesticides with the same active ingredient may be suitable. No endorsement is implied.
References
Amiri A., and Pandit LK. 2019. Evaluation of difenoconazole as postharvest fungicide to control ten major pathogens of pome fruit. APS-Pacific Division. Phytopathology 109-11-S3.8.
Ali EM., Mulvaney KA., Pandit LK., and Amiri A. 2018. Sensitivity of Phacidiopycnis spp. isolates from pome fruit to six pre- and postharvest fungicides. Plant Disease 102:533-539.
Amiri A., and Ali MDE. 2018. Prevalence and management of two emerging postharvest diseases of apple in Washington State. Proceedings of 11th IOBC/WPRS workshop on pome fruit diseases. Integrated Plant Protection in Fruit Crops, IOBC-WPRS Bulletin.
Amiri A., and Ali. MD.E. 2016. Prevalence of storage decays of apple: Lessons from the 2016 statewide survey.
Kim YK. and Xiao CL. 2006. A postharvest rot in apple caused by Phacidiopycnis washingtonensis. Plant Disease 90:1376-1381
Liu, Q., and Xiao, C. L. 2005. Influence of nutrient and environmental factors on conidial germination of Potebniamyces pyri. Phytopathology 95:572-580.
Xiao, C. L., and Boal, R. J. 2004. Prevalence and incidence of Phacidiopycnis rot in d’ Anjou pears in Washington State. Plant Disease 88:413-418.
Xiao, C. L., and Boal, R. J. 2005. Distribution of Potebniamyces pyri in the U.S. Pacific Northwest and its association with a canker and twig dieback disease of pear trees. Plant Disease 89:920-925.
Contact
 Dr. Achour Amiri,
Dr. Achour Amiri,
Postharvest Pathology
Tree Fruit Research and Extension Center
Wenatchee, WA
a.amiri@wsu.edu
509-293-8752
treefruit.wsu.edu articles may only be republished with prior author permission © Washington State University. Reprint articles with permission must include: Originally published by Washington State Tree Fruit Extension at treefruit.wsu.edu and a link to the original article.