Elizabeth H. Beers and Adrian T. Marshall, published online September 2017. Updated 2017 Betsy Beers.
Halyomorpha halys Stål (Heteroptera: Pentatomidae)

There are ca. 200 species of stink bugs in the US, 51 of which can be found in Washington. Some are beneficial (predators), but many are pests. Brown marmorated stink bug (Halyomorpha halys Stål) is an invasive pest from Asia, whose native range is China, Korea, and Japan. It was first found in the US in Pennsylvania in the mid-1990s, and in the Pacific Northwest (Portland) about 2004. Washington populations were first established in Vancouver (across the Columbia River from Portland), and have since spread north along the I-5 corridor. To date, brown marmorated stink bug has been recorded in 21 counties in Washington, with the majority of finds occurring in western Washington, along the Columbia River east to Walla Walla, and in Yakima.
While it has many features in common with native stink bug pests, damage potential from brown marmorated stink bug is considered higher due to its broad host range and ability to reproduce through two generations on tree fruits. In the eastern US, high levels of damage are associated with immigration from nearby deciduous wooded areas, which are absent in most parts of the semi-arid interior of PNW fruit growing regions. West of the Cascades, especially in the Willamette Valley of Oregon, brown marmorated stink bug is well established, and is now considered a commercial pest of hazelnuts. Although its potential for damage in semi-arid areas has not yet been determined, brown marmorated stink bug is classed as an agricultural and nuisance pest in the PNW.
BMSB Outreach
Wenatchee Valley Farmer’s Market, October 2017
BMSB finds in Washington (Map)
Fact Sheets and Press Coverage
If you see a BMSB, please send a photo, general location (inside or outside of house or car, backyard, park, etc.) and the address it was found to: tfrec.reportBMSB@wsu.edu
Parasitic Wasp Videos:


Hosts
Brown marmorated stink bug has a very broad host range, and is known to feed on a wide range of tree fruits, ornamentals, field crops, and fruiting vegetables. Apples are considered a high-risk crop, and while pears have been less studied in the eastern US, they also appear to be at risk. Peaches are also among the highest risk crops; however, the effect on cherries, apricots and plums has not been as well studied. The high percentage of the US apple, pear, and sweet cherry production in the western US (versus the eastern US where brown marmorated stink bug has been established longer) has made the risk level harder to gauge.
Life stages
Egg

The egg is typical for stink bugs, barrel shaped and light green, about 1.6 x 1.3 mm, with a circle of projections around the top. They are usually laid in clusters of 20-30 (average is 28). The egg clusters are laid on the underside of leaves of the host plant.
Nymphs
The nymphs develop through five instars. The first instar is 2.4 mm, black with yellowish red markings on the abdomen. The second instar is 3.7 mm, with a series of rectangular spots on the back of the abdomen (black on a reddish background), with the outer margin of the abdomen covered with black dashes. The antennae are reddish black, with a white band (3rd antennal segment). The third instar is 5.5 mm with similar markings, but bands on the legs are evident as well as the antennae. The fourth instar (8.5 mm) and fifth instar (12 mm) are similar in appearance to the previous instars only larger. By the third instar, the marginal dashes on the abdomen have white dots in the center of the outside edge, which are well developed by the fifth instar. All nymphal stages have a spiny/toothed margin on the front edge of the prothorax, unlike the adult which is smooth.
Adults
The adults are one of the larger stink bugs encountered in WA (12-17 mm). Adults have several key characteristics that distinguish it (apart from their large size): the alternating white bands on the antennae, and the smooth margin on the front edge of the prothorax (see photos). It also has alternating light and dark bands around margins of the abdomen; however, this feature is not unique to brown marmorated stink bug.
Brown marmorated stink bug is most likely to be confused with the other brown stink bugs, namely the consperse stink bug (Euchistus conspersus) and predatory (beneficial) stink bugs in the genus Brochymena (rough stink bugs). The rough stink bug has a coarsely toothed margin on the prothorax, with solid colored antennae. The consperse stink bug is much smaller, has a finely toothed margin on the prothorax, and lacks white banding on the antennae. Stink bugs in the genus Chlorochroa are also brown, but have smooth white margin on the prothorax and abdomen, and a white dot at the tip of the prothorax, where the forewings meet.
Life history
Research to date indicates 1-2 generations/year in the temperate part of its range. Although only preliminary information is available from eastern Washington, two generations are likely. Brown marmorated stink bugs overwinter as adults in sheltered areas. They are best known for their habit of overwintering in human habitations (houses, sheds, or barns), often in huge numbers; they are considered a household pest for this reason. They migrate gradually from their overwintering quarters as temperatures warm in the spring, and become reproductively active when the photoperiod increases to 12-13 hours (Nielsen et al., 2017). The adults disperse to host plants in spring and can begin laying eggs in orchards as early as May. The eggs develop to adults in 76 days (20°C), which then lay eggs and begin a second generation. This generation is often more numerous and damaging, especially as immigration from other hosts continues. Trapping data of adults in eastern WA indicates an early peak in mid-late May, and a second peak in July-August.
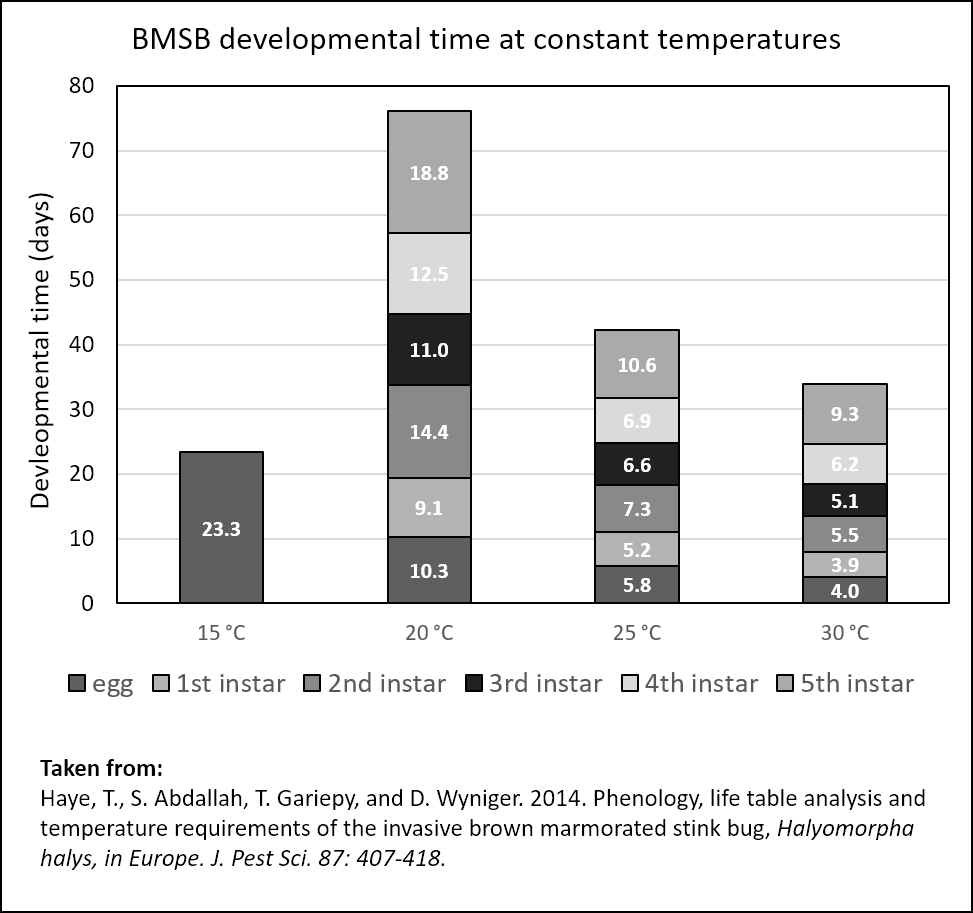 The eggs are laid on the underside of the leaves of host plants, and hatch after 6 to 26 days depending on temperature. The first instar nymphs remain clustered on the mass for several days, making them more vulnerable to predation. As the eggs disperse from the egg mass, they may move to and feed on several hosts before they complete development.
The eggs are laid on the underside of the leaves of host plants, and hatch after 6 to 26 days depending on temperature. The first instar nymphs remain clustered on the mass for several days, making them more vulnerable to predation. As the eggs disperse from the egg mass, they may move to and feed on several hosts before they complete development.
Adults also move around the landscape seeking out various hosts, and are capable of long-distance flight (Wiman et al., 2014), which facilitates local or regional dispersal. Long-distance dispersal (to new countries or regions) is facilitated by its habit of aggregating in inconspicuous areas of vehicles or shipped goods, and is the most likely explanation of the relatively fast invasion of North America and Europe.
Adults seek a sheltered place to spend the winter in late fall. The females enter reproductive diapause in the fall, and remain in this state until spring. Substantial overwintering mortality may occur if adults are exposed to extreme temperatures.
Damage
The damage is the same as that caused by native stink bugs, however, in the case of brown marmorated stink bug, the nymphs may cause considerable damage. The proboscis is inserted into the fruit, and salivary secretions form a stylet sheath, which is visible when the fruit is sliced. The entry point of the stylets on the surface is also visible as a tiny puncture. The injury may not be immediately apparent, but develops over time. In apples, a brownish depression may form around the feeding site. Damage may be visible at harvest, but becomes even more apparent during long-term storage.
Feeding sites on some crops (e.g., peaches, nectarines, apricot) may have gummosis extruding from the wound. In peaches, early feeding causes deformity and internal necrosis, and the appearance of damage may depend on the period of fruit maturity in which the damage occurred. Damage on cherries is characterized by a puncture on the surface, but internal damage has not been well studied.
Monitoring
There are two primary monitoring techniques: pheromone traps and beating tray samples. Unlike the sex pheromones of moth pests (which attracts males only), the pheromone of brown marmorated stink bug is an aggregation pheromone, which attracts males, females, and nymphs. The current most attractive pheromone is a two-part lure consisting of the aggregation pheromone (murgantiol) and a synergist, methyl decatrienoate (MDT), and is available from commercial pheromone vendors.
The lure can be deployed in a number of different trap styles, available from commercial vendors. The black pyramid trap is anchored on the ground with a jar on the top to house the lures and collect the insects. A killing agent (no-pest strip) will help retain the insects captured. This is considered a standard, proven technique, but rather expensive and cumbersome. Smaller traps, either staked to the ground or suspended in trees, may also catch brown marmorated stink bug, but generally catch fewer adults and nymphs. A simpler trap style, consisting of a clear sticky panel on a 4-foot wooden stake, is being tested, and may ultimately prove to be a more practical method even if captures are lower than in pyramid traps.
Beating trays are a traditional method of sampling for native stink bugs, and also work with brown marmorated stink bug. Sampling a favored host plant is recommended to find stink bugs. Because brown marmorated stink bugs may not enter traps (even though they are attracted to the vicinity) sampling plants close to a lure many enhance the chances of finding brown marmorated stink bug.
Brown marmorated stink bugs are also attracted to lights (including black lights), and this has been used for survey work. Use as a site-specific indicator of population pressure has not been implemented.
Biological control
Brown marmorated stink bug is attacked by predators and parasitoids, with the egg stage being the best studied and likely the most vulnerable target. Generalist chewing or sucking predators may account for substantial amounts of egg mortality, although finding the predator responsible is not obvious; gut content analysis may be helpful in identifying which predators are most successful. The predators implicated to date in brown marmorated stink bug egg predation include green lacewings, predatory mirids, cockroaches, katydids, earwigs, and ants. Predation on nymphs and adults has been less studied, but several crabronid wasps have been noted using brown marmorated stink bug nymphs to provision their nests. Spiders may be a significant mortality factor for overwintering adults inside dwellings. Large predators such as preying mantids and reduviids (wheel bugs) have been observed feeding on nymphs and adults. Carabid beetles, predatory stink bugs, and jumping spiders have been observed feeding on first instar nymphs on the egg mass.
Egg parasitoids are also a major potential source of biological control of brown marmorated stink bug. Unfortunately, the native US parasitoids (including wasps in the genera Telenomus, Trissolcus, Ooencyrtus and Anastatus) are not very successful in reproducing on brown marmorated stink bug eggs. They may, however, kill a number of eggs by ovipositing in them, even if the wasp fails to develop. Host feeding on the eggs by the female wasp may kill additional individuals; however, this is generally considered a ‘dead end’ for the parasitoid. Some successful emergence has been recorded, and the native species many eventually adapt to brown marmorated stink bug as a host. Anastatus reduvii is one of the most successful native egg parasitoid species for brown marmorated stinkbug in the PNW.
In contrast, egg parasitoids from brown marmorated stink bug’s native range in Asia are much better adapted, and thus more successful in attacking and reproducing on brown marmorated stink bug eggs. An exotic parasitoid, Trissolcus japonicus (the samurai wasp) has very high rate of parasitism of brown marmorated stink bug, and is considered the most promising candidate for importing into the US in a classical biological control program. Although several species of Asian parasitoids were imported in the late 2000s, they are still in quarantine awaiting approval for release. In the meantime, adventive populations (accidental introductions) have been found in the US, including several states in the mid-Atlantic area (Virginia to New York) . On the west coast, populations have been found in Washington (Milnes et al., 2016) and Oregon, thus this parasitoid appears to be on its way to widespread establishment. Release of reared or captured samurai wasps within state boundaries is ongoing in several regions, and will help facilitate spread.
There is less information on parasitoids of brown marmorated stink bug adults and nymphs, but so far only tachinid flies (some of which are known stink bug adult parasitoids) have been recorded to attack and emerge from brown marmorated stink bug, but instances are relatively rare.
Management
Most of the management recommendations on tree fruits originated from work done in the eastern US, where brown marmorated stink bug has been established longer. While homeowners in Washington have noted damage to backyard fruits, vegetables, and ornamentals, the only damage to commercial orchards has been to farms in the greater Vancouver, WA area. Adult brown marmorated stink bug have been detected in vineyards in the Walla Walla area, but no damage has been reported. At this point, commercial growers in eastern Washington are advised to be vigilant for brown marmorated stink bug, but not to apply prophylactic sprays.
In the eastern US, brown marmorated stink bug is notorious for both causing high levels of crop damage in certain years, and disrupting ongoing IPM programs. The influx of adults from May through September has provoked a cover spray program to prevent damage, resulting in outbreaks of secondary pests (Leskey et al., 2012). While severe crop losses have been reported since 2010, the insect pressure is not uniformly high in all areas or years. Brown marmorated stink bug is a landscape-level pest, moving from crop to crop to fulfill its nutritional needs. It is also considered a border pest as it invades orchard edges from nearby crops and wooded areas. Several current management strategies utilize this behavior by focusing efforts on the crop perimeter.
The potential for disruption of Washington’s IPM programs in pome fruit is severe if brown marmorated stink bug establishes at levels similar to the mid-Atlantic states. The majority of the insecticides recommended for control of brown marmorated stink bug are pyrethroids, although a few other groups (neonicotinyls and carbamates) also have active ingredients that are effective (Bergh et al., 2016). Tactics being investigated to reduce the non-target impact of insecticides include border applications to reduce total amounts of pesticides applied (Blaauw et al., 2015), attract-and-kill strategies (Morrison et al., 2016), and exclusion using net barriers. (Marshall and Beers, 2016). Establishing biological control (such as the samurai wasp) in urban areas may prevent build-up and spread to agricultural areas.
Materials available for apple
Extensive testing has been done in the eastern US where BMSB is more common. For material options visit Stop BSMB.
Use pesticides with care. Apply them only to plants, animals, or sites listed on the labels. When mixing and applying pesticides, follow all label precautions to protect yourself and others around you. It is a violation of the law to disregard label directions. If pesticides are spilled on skin or clothing, remove clothing and wash skin thoroughly. Store pesticides in their original containers and keep them out of the reach of children, pets, and livestock.
YOU ARE REQUIRED BY LAW TO FOLLOW THE LABEL. It is a legal document. Always read the label before using any pesticide. You, the grower, are responsible for safe pesticide use. Trade (brand) names are provided for your reference only. No discrimination is intended, and other pesticides with the same active ingredient may be suitable. No endorsement is implied.
References consulted
Abram, P. K., K. A. Hoelmer, A. Acebes-Doria, H. Andrews, E. H. Beers, C. Bergh, R. Bessin, D. J. Biddinger, P. Botch, M. L. Buffington, M. L. Cornelius, E. Costi, E. S. Delfosse, C. Dieckhoff, R. Dobson, Z. Donais, M. Grieshop, G. Hamilton, T. Haye, C. S. Hedstrom, M. Herlihy, M. Hoddle, C. Hooks, P. Jentch, N. K. Joshi, T. P. Kuhar, J. Lara, J. C. Lee, A. Legrand, T. C. Leskey, D. Lowenstein, L. Maistrello, C. R. Mathews, J. M. Milnes, W. R. Morrison III, A. L. Nielsen, E. C. Ogburn, C. H. Pickett, K. Poley, J. Pote, J. Radl, P. M. Shrewsbury, E. J. Talamas, L. Tavella, J. F. Walgenbach, R. A. Waterworth, D. C. Weber, C. Welty, and N. G. Wiman. 2017. Indigenous arthropod natural enemies of the invasive brown marmorated stink bug in North America and Europe. J. Pest Sci. 90: 1009-1020.
Anonymous. 2017. Trissolcus japonicus – the samurai wasp. http://www.stopbmsb.org/managing-bmsb/biological-control/trissolcus-japonicus-the-samurai-wasp/.
Bergh, C., A. Acebes-Doria, T. C. Leskey, R. Morrison, B. D. Short, G. Krawczyk, J. F. Walgenbach, A. M. Agnello, P. Jentsch, G. Hamilton, A. Nielsen, B. R. Blaauw, V. Walton, N. Wiman, C. H. Hedstrom, P. Shearer, and E. H. Beers. 2016. Integrated pest management for brown marmorated stink bug in orchard crops: A synopsis of what researchers have learned so far and management recommendations using an integrated approach. http://www.stopbmsb.org/stopBMSB/assets/File/BMSB-in-Orchard-Crops-English.pdf
Blaauw, B. R., D. Polk, and A. L. Nielsen. 2015. IPM-CPR for peaches: incorporating behaviorally-based methods to manage Halyomorpha halys and key pests in peach. . Pest Manag. Sci. 71: 1513-1522.
Hoebeke, E. R., and M. E. Carter. 2003. Halyomorpha halys (Stål) (Heteroptera: Pentatomidae): a polyphagous plant pest from Asia newly detected in North America. Proc. Entomol. Soc. Washington. 105: 225-237.
Leskey, T. C., G. C. Hamilton, A. L. Nielsen, D. F. Polk, C. Rodriguez-Saona, J. C. Bergh, D. A. Herbert, T. P. Kuhar, D. Pfeiffer, G. P. Dively, C. R. R. Hooks, M. J. Raupp, P. M. Shrewsbury, G. Krawczyk, P. W. Shearer, J. Whalen, C. Koplinka-Loehr, E. Myers, D. Inkley, K. A. Hoelmer, D.-H. Lee, and S. E. Wright. 2012. Pest status of the brown marmorated stink bug, Halyomorpha halys in the USA. Outlooks Pest Manag. 23: 218-226.
Marshall, A., and E. H. Beers. 2016. Non-target effects of net enclosures in apple orchards, In 90th Annual Orchard Pest & Disease Management Conference, 13-15 January, 2016, Portland Hilton, Portland, OR.
Milnes, J., N. G. Wiman, E. J. Talamas, J. F. Brunner, K. Hoelmer, M. L. Buffington, and E. H. Beers. 2016. Discovery of an exotic egg parasitoid of the brown marmorated stink bug, Halyomorpha halys (Stål) in the Pacific Northwest. Proc. Entomol. Soc. Washington. 118: 466-470.
Morrison III, W. R., A. N. Bryant, B. Poling, N. F. Quinn, and T. C. Leskey. 2017. Predation of Halyomorpha halys (Hemiptera: Pentatomidae) from web-building spiders associated with anthropogenic dwellings. J. Ins. Behav.: 1-16.
Morrison, W. R. I., J. P. Cullum, and T. C. Leskey. 2015. Evaluation of trap designs and deployment strategies for capturing Halyomorpha halys (Hemiptera: Pentatomidae). J. Econ. Entomol. 108: 1683-1692.
Morrison, W. R. I., D.-H. Lee, B. D. Short, A. Khrimian, and T. C. Leskey. 2016. Establishing the behavioral basis for an attract-and-kill strategy to manage the invasive Halyomorpha halys in apple orchards. J. Pest Sci. 89: 81-96.
Nielsen, A. L., S. Fleischer, G. C. Hamilton, T. Hancock, G. Krawczyk, J. C. Lee, E. Ogburn, J. M. Pote, A. Raudenbush, A. Rucker, M. Saunders, V. P. Skillman, J. Sullivan, J. Timer, J. Walgenbach, N. G. Wiman, and T. C. Leskey. 2017. Phenology of brown marmorated stink bug described using female reproductive development. Ecol. Evol.: 1-11.
Oregon Department of Agriculture. 2010. Pest Alert: Brown marmorated stink bug.
Wiman, N. G., V. M. Walton, P. W. Shearer, S. I. Rondon, and J. C. Lee. 2014. Factors affecting flight capacity of brown marmorated stink bug, Halyomorpha halys (Hemiptera: Pentatomidae). J. Pest Sci. 88: 37-47.
Zack, R. S., P. J. Landolt, and J. E. Munyaneza. 2012. The Stink Bugs (Hemiptera: Heteroptera: Pentatomidae) of Washington State. Great Lakes Entomologist. 45: 251-262.
Zhang, J., F. Zhang, T. Gariepy, P. Mason, D. Gillespie, E. Talamas, and T. Haye. 2017. Seasonal parasitism and host specificity of Trissolcus japonicus in northern China. J. Pest Sci.: 1-15.
Contacts
Elizabeth Beers
Department of Entomology
Washington State University
Tree Fruit Research & Extension Center
Wenatchee, WA 98801
Phone: 509.293.8755
CAHNRS Tree Fruit Entomology/Elizabeth Beers website
email: ebeers@wsu.edu
Adrian Marshall
Ph.D. student
Washington State University
Tree Fruit Research & Extension Center
1100 N Western Ave.,
Wenatchee, WA 98801
Phone: 509.293-8756
email: atmarshall@wsu.edu
Articles from the Tree Fruit website may only be republished with prior author permission © Washington State University. Reprint articles with permission must include: Originally published by Washington State Tree Fruit Extension Fruit Matters at treefruit.wsu.edu and a link to the original article.









