The Pacific Northwest Sweet Cherry Breeding Program was re-established in 2004 to develop superior new cultivars for the PNW Sweet Cherry industries. Extensive breeding resources, including a diverse germplasm base, with a continuum of genetically improved plant material from parents to seedlings and advanced selections, has been established to start a new stage of the WSU Cherry Breeding Program.
The Pacific Northwest Sweet Cherry Breeding Program was re-established in 2004 to develop superior new cultivars for the PNW Sweet Cherry industries. Extensive breeding resources, including a diverse germplasm base, with a continuum of genetically improved plant material from parents to seedlings and advanced selections, has been established to start a new stage of the WSU Cherry Breeding Program.
In the last two years, Dr. Cameron Peace (WSU, Genetics) and Bernardita Sallato (WSU Horticulturist) have been preparing for an incoming new permanent breeder by streamlining the performance evaluation system while raising and keeping healthy trees. The industry members have been fundamental by guiding and advising through the Cherry Breeding Program Advisory Committee (BPAC) and the Washington Tree Fruit Research Commission (WTFRC).
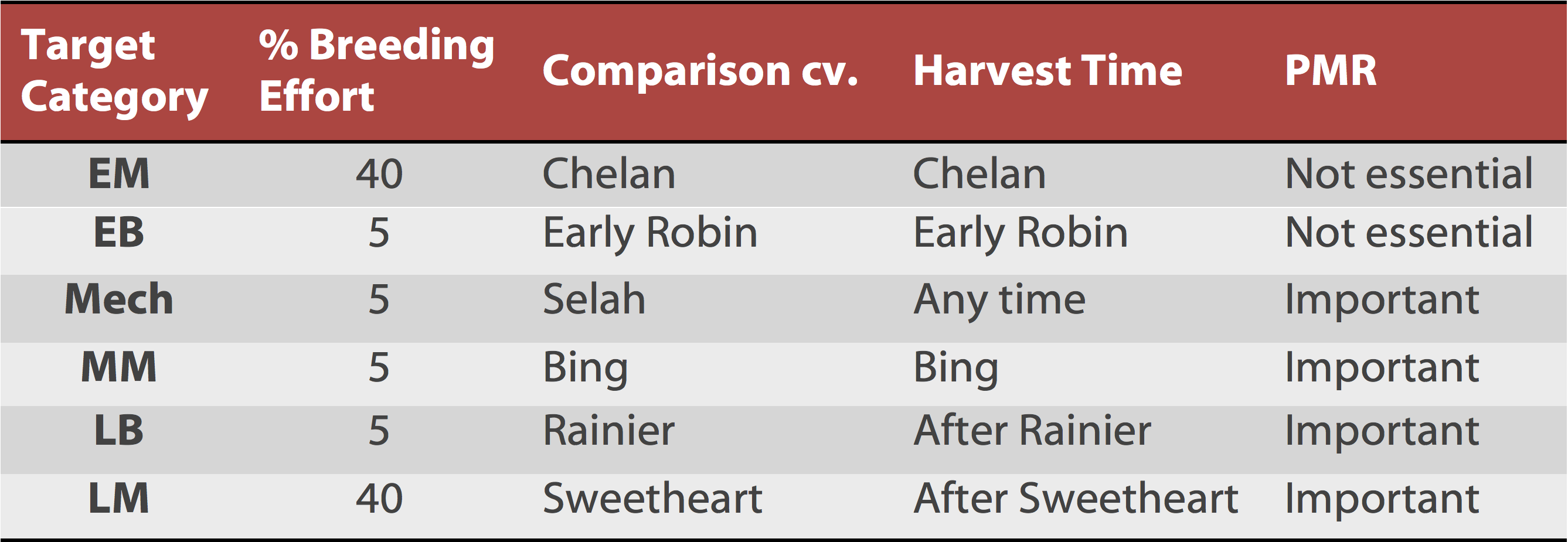
The program focusses in 5 main ideotypes with different efforts allocated according to the industry priorities (Table 1).
Table 1. Sweet cherry breeding selection parameters and descriptors for the Pacific Northwest region.
Keys for table:
EM = Early season Mahogany: ≦ Chelan timing
EB = Early season Blush: ≦ Early Robin timing
Mech = Mechanical
MSM = Mid-season Mahogany: Bing timing
LSB = Late season Blush: > Rainier timing
LSM = Late Season Mahogany: ≥ Sweetheart timing
PMR = Powder mildew resistance
The program has 3 phases. Phase 1 includes making new crosses, planting own-rooted seedlings and single tree performance evaluation. Phase 2 includes replicated trials of grafted advanced selections at 3 locations representing each market timing: Early site (Pasco); Middle season site (Prosser); and a late site (Wenatchee) in WA, and one site in OR (MCAREC-Hood River). Phase 3 corresponds to the replicated trials of elite selections on grower-cooperator orchards in WA and OR prior to release. Currently, there are no advanced selections for the late-season market class nor Phase 3 elite selections.
During early 2017, the WSU team with BPAC advice, discarded nine advanced selections (out of a total of 24 going into 2017) considered not promising enough in fruit quality and/or productivity or that exhibited fatal flaws, based on established PNW industry needs and priorities.
 DNA profiling has been used to improve breeding efficiency. All parent material and current Phase 2 selections have been analyzed for whole-genome DNA supported by the RosBREED project. Genetic tests for self-fertility and fruit size ensure that only parents with a high probability of producing large fruited and self-fruitful progenies were used in crosses in the past. The tests also helped to eliminate inferior seedlings so only seedlings that have favorable alleles for desired traits were planted in what it is now the Phase 1 seedlings. The marker-locus-trait (MLT) tags will be deployed once very reliable, robust genetic tests are available.
DNA profiling has been used to improve breeding efficiency. All parent material and current Phase 2 selections have been analyzed for whole-genome DNA supported by the RosBREED project. Genetic tests for self-fertility and fruit size ensure that only parents with a high probability of producing large fruited and self-fruitful progenies were used in crosses in the past. The tests also helped to eliminate inferior seedlings so only seedlings that have favorable alleles for desired traits were planted in what it is now the Phase 1 seedlings. The marker-locus-trait (MLT) tags will be deployed once very reliable, robust genetic tests are available.
Current Focus of the Cherry Breeding Program
The WSU sweet cherry breeding program continues to concentrate on finding improved cherry cultivars that will benefit both growers and consumers. Greater efforts are directed to developing early mahogany and late mahogany type cherries, with PMR especially in the mid-season and late varieties, and self-fruitfulness in all market classes. The focus for selection is exceptional fruit quality at harvest and postharvest for each market class, which includes size, firmness, good flavor, and appealing fruit. Additional characteristics are evaluated either to efficiently discard selections with fatal flaws or select for specific markets classes, for example, mechanical harvesting, resistance to cracking, heat tolerance, crop load, and ripening variability.
Powdery mildew resistance (PMR) is one of the most demanded traits required for the mid-season and late varieties for the PNW Sweet Cherry industry. During 2017, in collaboration with Gary Grove and Claudia Probst, a reliable and efficient assay for revealing the genetic potential for fruit powdery mildew (PM) resistance was developed and is routinely used in the PNWSCBP. Resistant and susceptible breeding individuals have been successfully discriminated for both fruit and foliar PM infection. Although this project goal is ongoing, preliminary results indicate that the genetic source for resistance from PMR-1 (inherited from ‘Moreau’) is the same as that from the small-fruited Mildew-Immune Mazzards. This source of resistance appears to be common within the PNW sweet cherry breeding program due to the extensive use of sources carrying the resistance allele for at least the last 10 years of crossing.
 A total of 12 selections were evaluated in Phase 2 in 2017. Several promising selections stood out: R1, R3, R17, R19, and especially R29. R19 is Early Mahogany, 4 days after Chelan and 10 days before Bing in both 2016 and 2017; while the other four selections are Mid-season Mahogany from approximately one week before to four days after Bing timing. Selection R29 showed particularly promising characteristics compared to its standard Bing, having substantially larger and firmer fruit (average 14.8 grs, 32 mm of fruit size and 321 g/mm of firmness). Selection R29 has a sweet flavor and an attractive appearance (shiny luster and good color development) at harvest and post-storage. However, it has low acidity compared to Bing. In 2018 we expect enough crop for R29 in Pasco for a full harvest and storage evaluation.
A total of 12 selections were evaluated in Phase 2 in 2017. Several promising selections stood out: R1, R3, R17, R19, and especially R29. R19 is Early Mahogany, 4 days after Chelan and 10 days before Bing in both 2016 and 2017; while the other four selections are Mid-season Mahogany from approximately one week before to four days after Bing timing. Selection R29 showed particularly promising characteristics compared to its standard Bing, having substantially larger and firmer fruit (average 14.8 grs, 32 mm of fruit size and 321 g/mm of firmness). Selection R29 has a sweet flavor and an attractive appearance (shiny luster and good color development) at harvest and post-storage. However, it has low acidity compared to Bing. In 2018 we expect enough crop for R29 in Pasco for a full harvest and storage evaluation.
The WSU Breeding program also continues to preserve germplasm with good horticultural practices including monitoring irrigation, standard sprays, plant nutrition and disease management, virus routinely testing, training and pruning according to the specifics needs in each stage of the program. As a result, during 2017 more than 50% of the cherry breeding footprint was reduced.
Additional Resources
Maintenance and evaluation of sweet cherry breeding program germplasm. G. Grove, Final report WTFRC, 2017.
Breeding the new cherry, S. Dininny, Good Fruit Grower, May 2016
Breeding better rootstocks, S. Dininny, Good Fruit Grower, May 27, 2016
New cherry varieties wanted urgently, G. Warner, GFG, May 2015.
Early cherry is on fast track, G. Warner, GFG, May 2015.